
These 6 protons add up to a +6 charge, but carbon also has 6 electrons too, which supply a -6 charge, leading to no overall charge. 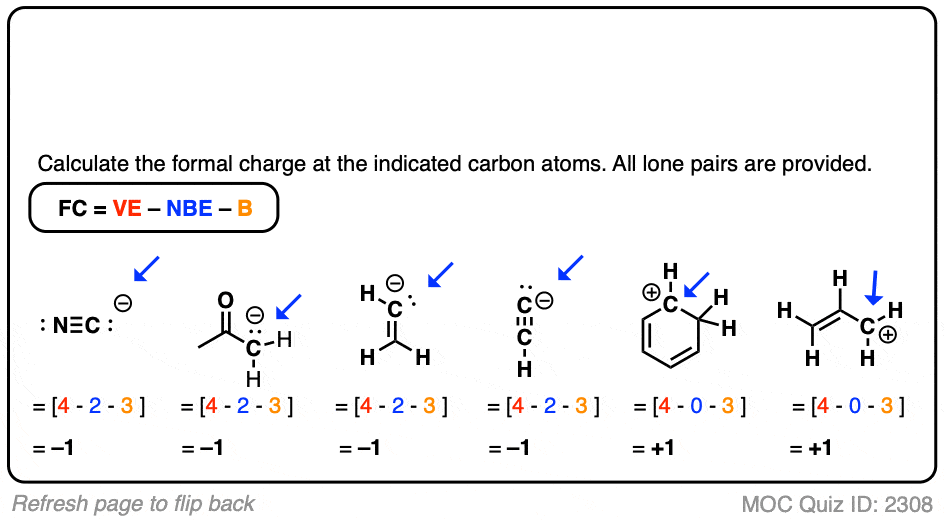
that’s why, its charge is 4⁺.įor example, carbon has 6 protons, we know this because the atomic number (lower number on the periodic table) for carbon is 6. it shares its valence electrons with other atoms and forms covalent bonds. carbon can lose or gain 4 electrons in order to complete its octet. Ĭarbon atom has 4 electrons in its valence shell or its outermost shell. So the total charge of the nucleus of a carbon atom is +6.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
